Everything is made of atoms
To understand electricity, it helps to know a little about atoms. Atoms are the tiny building blocks that make up everything in the universe—every star, every tree, every animal, and humans, too. Air and water are also made of atoms. Atoms are incredibly small; millions of them could fit on the head of a pin.
Atoms are made of even smaller particles
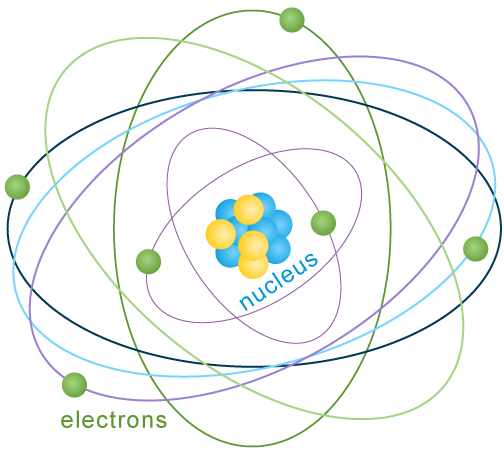
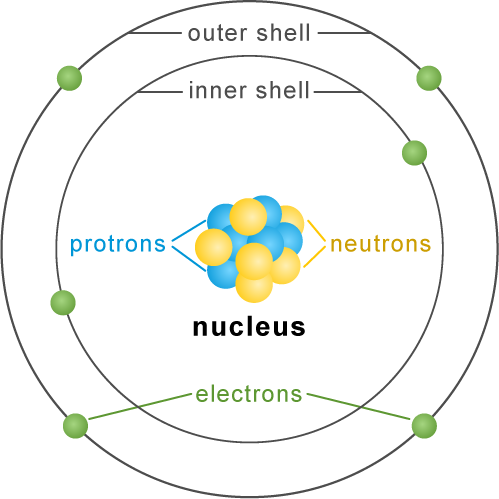
Each atom has a center called the nucleus. The nucleus contains particles called protons and neutrons. Even smaller particles called electrons spin around the nucleus in layers, or shells.
Imagine if the nucleus of an atom was the size of a tennis ball, the entire atom would be about the size of a large sports stadium (about 1,450 feet across). Atoms are mostly empty space.
Parts of an atom
- Nucleus: The center of the atom, containing protons and neutrons
- Protons: Particles with a positive (+) electrical charge
- Neutrons: Particles with no electrical charge
- Electrons: Particles with a negative (-) electrical charge that orbit the nucleus
Protons and electrons are attracted to each other because they have opposite charges (positive attracts negative). Generally, an atom has the same number of protons and electrons, so the positive charge of the protons balances out the negative charge of the electrons, making the atom stable. The neutrons carry no charge, and their number can vary.
Electrons stay in their shells because of this electrical force. They constantly move and try to stay as far apart as possible within their shell.
Protons determine the type of atom
The number of protons in an atom determines the kind of atom, or element, it is. An element is a substance consisting of one type of atom. The Periodic Table of Elements shows elements with their atomic numbers—the number of protons each has. For example, every atom of hydrogen (H) has one proton and every atom of carbon (C) has six protons.
Electricity is the movement of electrons between atoms
Electrons usually remain a constant distance from the atom's nucleus in precise shells. The shell closest to the nucleus can hold two electrons, the next shell can hold up to eight, and the outer shells can hold even more.
The electrons in the shells closest to the nucleus are pulled strongly toward the protons. Sometimes, the electrons in an atom's outermost shells have a weaker pull toward the protons. These outer electrons can be pushed out of their orbits and move from one atom to another. These shifting electrons are what we call electricity.
Static electricity exists in nature
You’ve probably experienced static electricity. Have you ever felt a shock when you touched an object after walking across a carpet? You felt a stream of electrons jumping to you from that object, which we call static electricity.
Have you ever made your hair stand straight up by rubbing a balloon on it? If so, you moved electrons into your hair from the balloon. The electrons all have the same electrical charge and push away from each other (like charges repel like), making your hair stand on end.
Lightning is also a form of electricity. Lightning is electrons moving from one cloud to another or electrons jumping from a cloud to the ground.