Hydrogen basics
What is hydrogen?

Source: NASA (public domain)
Did you know?
Hydrogen is the lightest element. Hydrogen is a gas at normal temperature and pressure, but hydrogen condenses to a liquid at minus 423 degrees Fahrenheit (minus 253 degrees Celsius).
Hydrogen is the simplest element. Each atom of hydrogen has only one proton. Hydrogen is also the most abundant element in the universe. Stars such as the sun consist mostly of hydrogen. The sun is essentially a giant ball of hydrogen and helium gases.
Hydrogen occurs naturally on earth only in compound form with other elements in liquids, gases, or solids. Hydrogen combined with oxygen is water (H2O). Hydrogen combined with carbon forms different compounds—or hydrocarbons—found in natural gas, coal, and petroleum.
Hydrogen is an energy carrier
Energy carriers transport energy in a usable form from one place to another. Hydrogen, like electricity, is an energy carrier that must be obtained from another substance. Hydrogen can be separated from a variety of sources including water, fossil fuels, or biomass and used as a source of energy or fuel. Hydrogen—in gaseous or liquid form—has the highest energy content of any common fuel by weight (about three times more than gasoline) but has the lowest energy content by volume. Even liquid hydrogen has only about one-fourth of the energy content as a comparable volume of gasoline.
It takes more energy to produce hydrogen (by separating it from other elements in molecules) than hydrogen provides when it is converted to useful energy. However, hydrogen is useful as an energy source/fuel because it has a high energy content per unit of weight, which is why it is used as a rocket fuel and in fuel cells to produce electricity on some spacecraft. Hydrogen is not widely used as a fuel now, but it has the potential for greater use in the future.
How is hydrogen produced?
To produce hydrogen, it must be separated from the other elements in the molecules where it occurs. There are many different sources of hydrogen and ways for producing it for use as a fuel. The two most common methods for producing hydrogen are steam reforming and electrolysis (splitting water with electricity). esearchers are exploring other hydrogen production methods, or pathways.
Steam reforming is a widely used method of hydrogen production
Most of U.S. hydrogen production is by steam methane-reforming. Commercial hydrogen producers and petroleum refineries use steam-methane reforming to separate hydrogen atoms from carbon atoms in methane (CH4). The steam reforming process results in some carbon dioxide emissions.
Industrial facilities and petroleum refineries primarily use natural gas as the methane source for hydrogen production. Several fuel cell power plants in the United States treat and use landfill gas (biogas) as hydrogen source. Biofuels and petroleum fuels are also potential hydrogen sources.
Electrolysis uses electricity
Electrolysis is a process that splits hydrogen from water using an electric current. Electrolysis is commonly used in high school science classes to demonstrate chemical reactions and hydrogen production. On a large, commercial scale, the process may be referred to as power-to-gas, where power is electricity and hydrogen is gas. Electrolysis itself does not produce any byproducts or emissions other than hydrogen and oxygen. The electricity for electrolysis is currently provided by the electric power grid, which is supplied with a mix of renewable sources, nuclear energy, and fossil fuels.
Other methods of producing hydrogen
Research is underway to develop other ways to produce hydrogen, including:
- Using microbes that use light to make hydrogen
- Converting biomass into gas or liquids and separating the hydrogen
- Using solar energy technologies to split hydrogen from water molecules
Clean hydrogen production
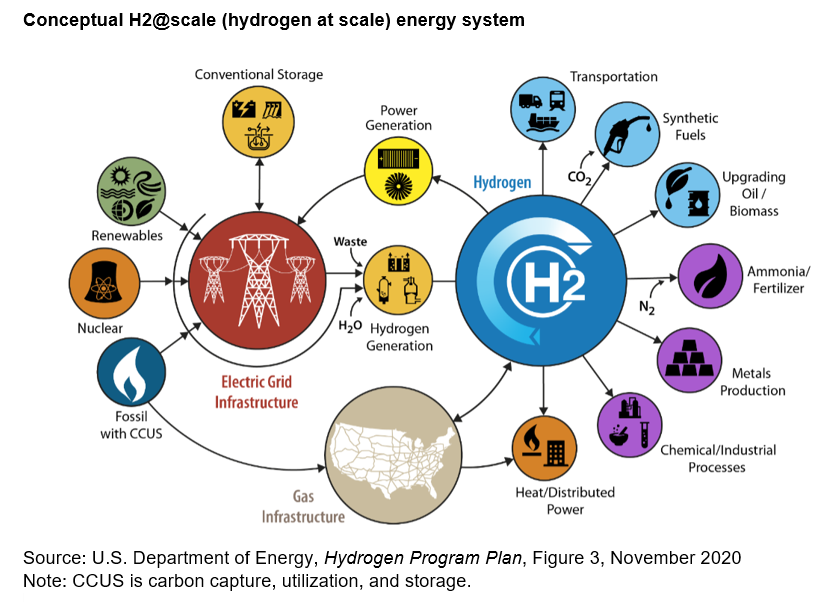
The U.S. Department of Energy (DOE) supports source-neutral hydrogen production pathways and does not use a color code that is based on the hydrogen source, production technology, and carbon capture, which some producers, marketers, and other organizations use to categorize hydrogen. DOE's Hydrogen Program includes a number of programs by participating DOE program offices for clean hydrogen production.
Use of hydrogen

Source: National Aeronautics and Space Administration (NASA) (public domain)
Source: Adapted from the National Energy Education Project (public domain)

Source: Wikimedia Commons
Hydrogen is currently used in industrial processes, as rocket fuel, and in fuel cells for electricity generation and powering vehicles. The National Aeronautics and Space Administration (NASA) has used hydrogen since the 1950s for space exploration. Operators of several natural gas-fired power plants are exploring the use of hydrogen to supplement or replace natural gas. Hydrogen has the potential for effectively storing energy for electricity generation.
Hydrogen is used in industrial processes
Nearly all of the hydrogen consumed in the United States is used by industry for refining petroleum, treating metals, producing fertilizer and other chemicals, and processing foods. U.S. petroleum refineries use hydrogen to lower the sulfur content of fuels. Biofuel producers also use hydrogen to produce hydrotreated vegetable oil (HVO) for use as renewable diesel.
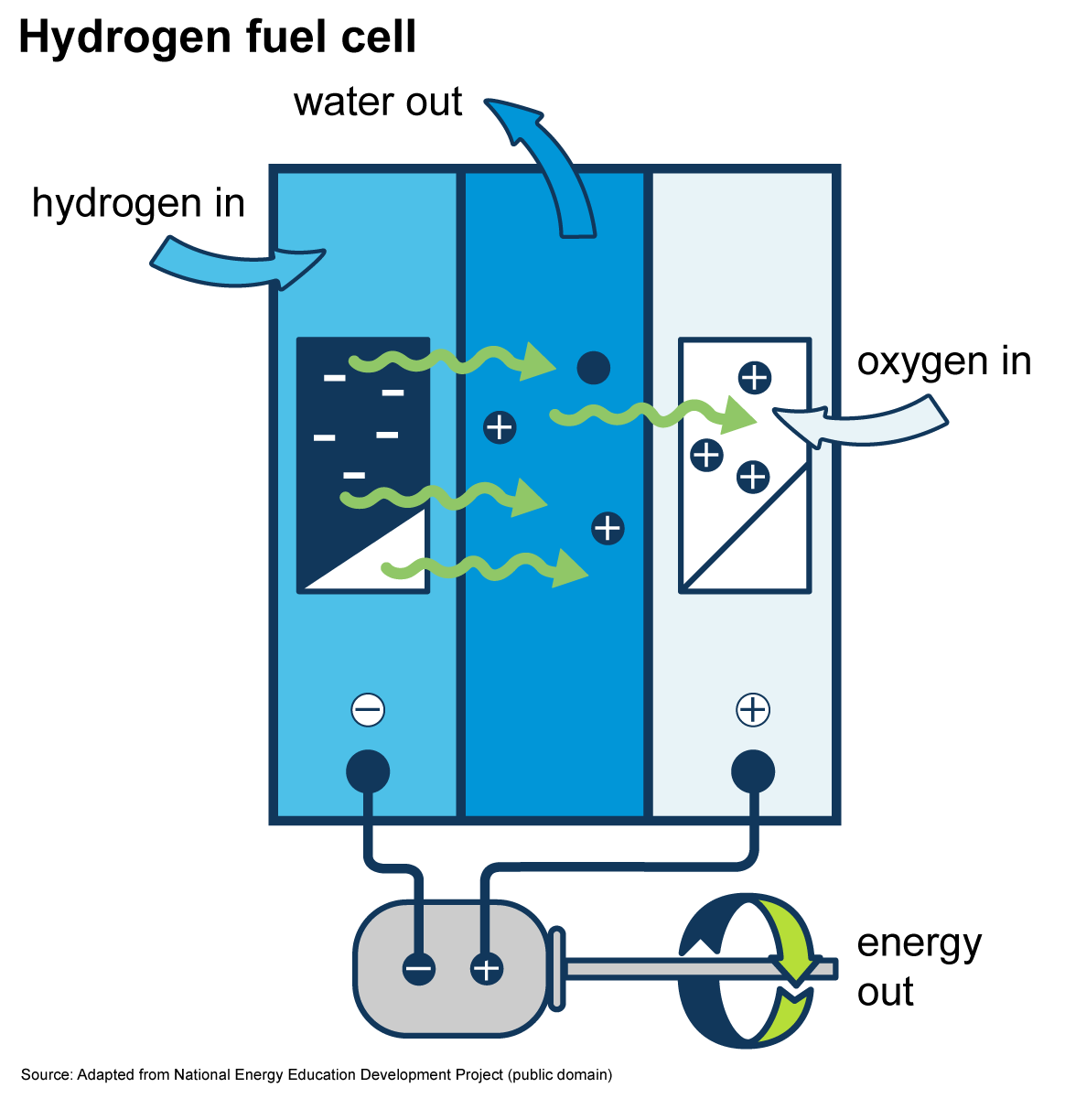
Hydrogen fuel cells produce electricity
Hydrogen fuel cells produce electricity by combining hydrogen and oxygen atoms. The hydrogen reacts with oxygen across an electrochemical cell—similar to a battery—to produce electricity, water, and small amounts of heat.
Hydrogen fuel cells are currently used to power the electrical systems on spacecraft and to supply electricity on earth. Small fuel cells have been developed to power electronic devices, such as laptop computers and cell phones. Several vehicle manufacturers have developed fuel cells to power vehicles. Fuel cells can potentially provide electricity for emergency power in buildings and for remote locations that are not connected to electric power lines.
Fuel cell power plants
As of the end of December 2022, the United States had about 205 operating fuel cell electric power generators at 147 facilities with about 350 megawatts (MW) of total nameplate electricity generation capacity. The nameplate capacities range from the largest single-fuel cell, with about 17 MW capacity—the Bridgeport Fuel Cell, LLC in Connecticut—to 10 fuel cells each with 0.1 MW capacity at the California Institute of Technology. The majority of all operating fuel cells use pipeline natural gas as the hydrogen source, but one uses landfill gas and four use biogas from wastewater treatment.
Fuel cells for vehicles
Hydrogen is considered an alternative vehicle fuel under the Energy Policy Act of 1992. The interest in hydrogen as an alternative transportation fuel stems primarily from its potential to power fuel cells in zero-emission vehicles (vehicles with no emissions of air pollutants). A fuel cell may be two to three times more efficient than an internal combustion engine running on gasoline. Several vehicle manufacturers have hydrogen fuel cell-vehicles available in California and Hawaii. A few test vehicles are available to organizations with access to hydrogen-fueling stations.
The high cost of fuel cells and the small number of hydrogen fueling stations have limited the number of hydrogen-fueled vehicles in use today. Production of hydrogen-fueled vehicles is limited because people won't buy those vehicles if hydrogen refueling stations are not easily accessible, and companies won't build refueling stations if they don't have customers with hydrogen-fueled vehicles. The United States has about 56 hydrogen-vehicle-fueling stations, all of which are in California.
Hydrogen can be burned for electricity generation and heating
Hydrogen combustion for electricity generation and for space heating in buildings are potential uses of pure hydrogen or hydrogen-rich blends with natural gas. However, hydrogen and hydrogen-blends in existing natural gas pipelines, electrity generators, and space-heating equipment poses a number of challenges. More research, development, and demonstration (RD&D) is needed before hydrogen will qualify for use in power plants. Several power plants in the United States have announced plans to operate on a natural gas-hydrogen fuel mixture in combustion-gas turbines. One example is the Long Ridge Energy Generation Project in Ohio. RD&D is also needed for using hydrogen and hydrogen–natural gas blends in heating appliances.
Hydrogen can be used for energy storage
Hydrogen storage is an important technology to enable hydrogen use across the U.S. economy. Hydrogen may be stored as a:
- Gas–Hydrogen can be stored as a gas in large volumes in natural geological formations–salt caverns, lined hard rock caverns, depleted oil and natural gas fields, and aquifers. Gaseous hydrogen may also be stored in relatively smaller volumes in pressurized, stationary or portable tanks, and in dedicated hydrogen gas pipeline infrastructure. Gaseous storage is currently the most common and the most likely option for expanding hydrogen storage for most uses of hydrogen as an energy source.
- Liquid–Hydrogen can be liquefied by cooling it to below −423oF (−253oC). The liquefied hydrogen can be stored in super-cooled (cryogenic) tanks for transportation applications in fuel cell vehicles or directly as fuel in truck, rail, marine, and rocket engines. NASA has the two largest liquid hydrogen storage tanks in the world. Hydrogen liquefaction and cyrogenic liquid storage is an energy intensive and expensive process.
Hydrogen could help to reduce carbon dioxide emissions from electricity generation by storing energy produced with renewable energy for use over days or weeks. Hydrogen could be produced with renewable resources when renewable energy production is high and could be stored to generate electricity when renewable resources are limited and electricity demand is high. One example is the Advanced Clean Energy Storage project in Utah, which plans to store large volumes of gaseous hydrogen produced from renewable resources for long-term seasonal energy storage.